We will be at DCAT Week 2026!
23-26th March 2026 in NYC
Don’t miss our presentations at PODD and CPHI!
- PODD
- Monday, October 27, 2025 – 05:30 PM - 05:45 PM
A Novel Approach to Modelling Injector Performance and Break-Loose Extrusion Force Data
Presenter: Tom Oakley, Springboard, co-presenting with Deep Bhattacharya, Pfizer
- Monday, October 27, 2025 – 05:30 PM - 05:45 PM
- CPHI
- Wednesday, October 29, 2025 – 11:35 AM - 12:05 PM
Rethinking Injector Mechanics – A New Approach to BLEF-Based Modelling
Speakers: Tom Oakley, Springboard, co-presenting with Jay Sayed, Pfizer
- Wednesday, October 29, 2025 – 11:35 AM - 12:05 PM
-
- Wednesday, October 29, 2025 – 12:05 PM - 12:20 PM
Modelling to Market – Enabling Smarter, Faster Injector Development
Speaker: Alex Vasiev, Springboard
- Wednesday, October 29, 2025 – 12:05 PM - 12:20 PM
Meet our experts at the conferences
Feel free to reach out to our sales representatives to arrange a meeting in advance.
Click on the images to send an email.
We are happy to welcome you at our DCAT Suite
in the Lotte NY Palace #2002.
Come meet us at New York City’s flagship annual business development event for global pharmaceutical manufacturing companies and find out how we can support you throughout your entire development and manufacturing value chain.

Stefan Verheyden
CEO
S.Verheyden@sanner-group.com

Christian Classen
CSO
C.Classen@sanner-group.com

Ted Mosler
CTO
tmosler@gilero.com

Geoff Jones
VP Sales, CDMO – Europe & North America
g.jones@sanner-group.com

Chuck Crandley
Associate VP Sales, CDMO
ccrandley@gilero.com
Sanner Group - Who we are
![]() Our Vision
Our Vision
Protecting Health.
![]() Our Mission
Our Mission
To partner with healthcare companies to develop and manufacture products globally.
![]() Our values
Our values
- Reliable
- Agile
- Collaborative
- Transparent
- Sustainable

The value we add to you
As a CDMO, we provide a full spectrum of services, from the initial design and development phases to mass production of everything from simple, single-use consumables to complex electromechanical devices.
With more than 60 years of experience in providing physical and chemical stability packaging for moisture sensitive drug products, we offer desiccant solutions for medical and diagnostic devices.
Our agility, scalability and substantial production capacity allows us to cater to a wide range of specific customer needs—whether for small production runs or high-volume manufacturing.
Supported by our state-of-the-art production facilities across Europe, Asia, and North America, Sanner is a strong partner of choice to support our clients to bring medical devices to their local markets efficiently.
What makes the difference
“At Sanner, we turn complex device ideas into scalable solutions.”
With a pool of 150 specialized engineers, we assemble exactly the right team for your development challenge.
Our global footprint across three continents seamlessly connects development and manufacturing—reducing risk and accelerating time to market.
And as an agile, customer-focused partner, we bring available capacity, fast decision-making, and a solution-oriented mindset to move your project forward.
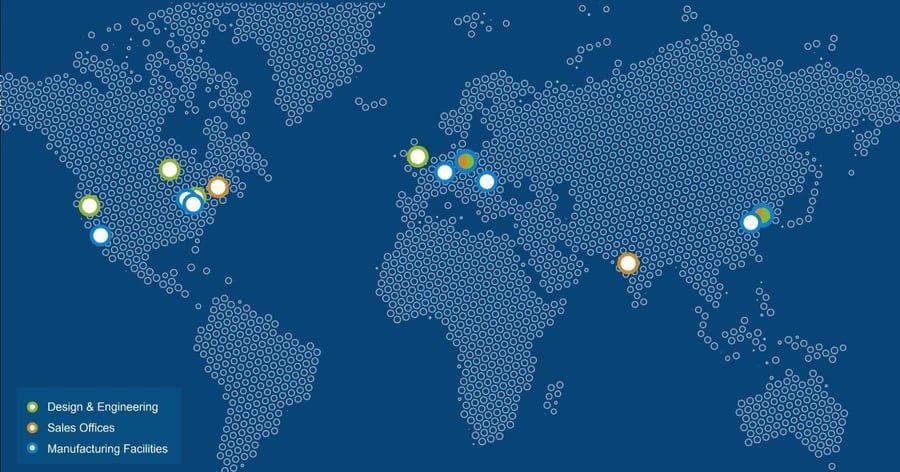
We offer worldwide production capacity and device development services
Sanner operates a global manufacturing network with a total production space of 28,500 sqm, including 11,000 sqm of cleanroom space (Class 7/8). Our Design Centers of Excellence, Springboard and Gilero, are strategically located in the UK and across the US. Our manufacturing facilities are based in the US, Asia and Europe.
How we can support you as a global medical device CDMO
- Capacity for small-batch prototypes and scaling to full production. Our facilities are equipped to handle a wide range of production needs:
- High precision fully electric thermoplastic injection molding capable of handling small shot weights (around 0.1g) up to 64-cavity molds (max. 350T), including multi-K molding with thermoplastic elastomers (TPE).
- Manual assembly up to fully automated assembly processes, incorporating mechatronic components for enhanced efficiency and precision.
- End of line packaging for sub-assemblies or finished goods
- Sterilization through trusted external partners, utilizing validated processes to ensure compliance with industry standards.
- Our experience in successfully transferring innovations to production across the EU, US, and China speaks to our ability to bring cutting-edge technologies to global markets. All phases of device development can be supported from our Design and Development Centers of Excellence in both the US and EU.
- Our design transfer processes enable the smoother path to market. We offer end-to-end thinking from development to manufacturing all under one roof to de-risk your project and for a better time to market.
- Our reliability and collaborative working attitude support you in any stage of your project.
Quality control processes and compliance with ISO 13485
Sanner Group facilities are ISO 13485 certified for Design & Development and Manufacturing respectively. Over 250 ISO, AAMI/ANSI standards are used routinely. Our streamlined quality system satisfies the FDA requirements for both the device quality system regulations and the part 210/211 cGMP regulations.