Custom Ophthalmic Drug Delivery Devices
From early development through commercialization, we help bring ophthalmic drug-delivery devices
to market, including implant injectors, liquid injectors, eye droppers, and topical applicators.
Don’t miss our presentations at PODD and CPHI!
- PODD
- Monday, October 27, 2025 – 05:30 PM - 05:45 PM
A Novel Approach to Modelling Injector Performance and Break-Loose Extrusion Force Data
Presenter: Tom Oakley, Springboard, co-presenting with Deep Bhattacharya, Pfizer
- Monday, October 27, 2025 – 05:30 PM - 05:45 PM
- CPHI
- Wednesday, October 29, 2025 – 11:35 AM - 12:05 PM
Rethinking Injector Mechanics – A New Approach to BLEF-Based Modelling
Speakers: Tom Oakley, Springboard, co-presenting with Jay Sayed, Pfizer
- Wednesday, October 29, 2025 – 11:35 AM - 12:05 PM
-
- Wednesday, October 29, 2025 – 12:05 PM - 12:20 PM
Modelling to Market – Enabling Smarter, Faster Injector Development
Speaker: Alex Vasiev, Springboard
- Wednesday, October 29, 2025 – 12:05 PM - 12:20 PM
Meet our experts at the conferences
Feel free to reach out to our sales representatives to arrange a meeting in advance.
Click on the images to send an email.
Experts in Designing and Manufacturing Solid and Liquid Dose Delivery Devices
Our team brings experience in designing devices across a range of active ingredients, including hydrogels, sustained-release solids, and small-molecule drug liquids.


Human Centered Design
Proven expertise in injection device design that balances patient outcomes with manufacturability.

Device Development
Holistic combination-product support from an expert device partner, ensuring device and program success.

Scalable Manufacturing
In-house clinical-grade component and final assembly manufacturing to support increasing production volumes.
Your expert partner for developing and manufacturing
custom ophthalmic drug delivery devices
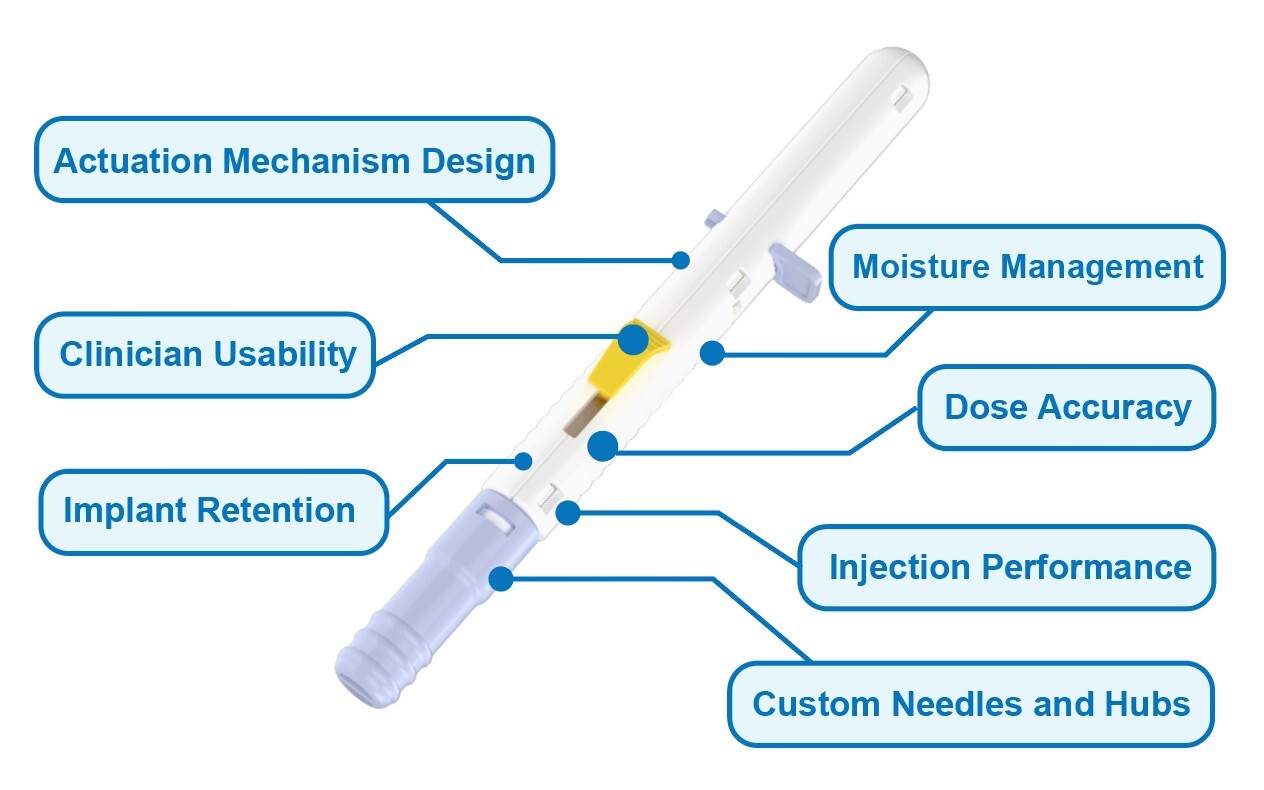
Sanner’s Robust Approach to Ocular Injection Devices
Our specialized expertise in ophthalmic injection device development supports ocular therapeutics programs from pre-clinical stages through commercialization.
With proven know-how in bringing ophthalmic injectors to market, we serve as a reliable device partner throughout your product’s journey to market success. We collaborate closely with your existing pharmaceutical manufacturing team to ensure seamless integration of drug and device as your clinical program progresses.
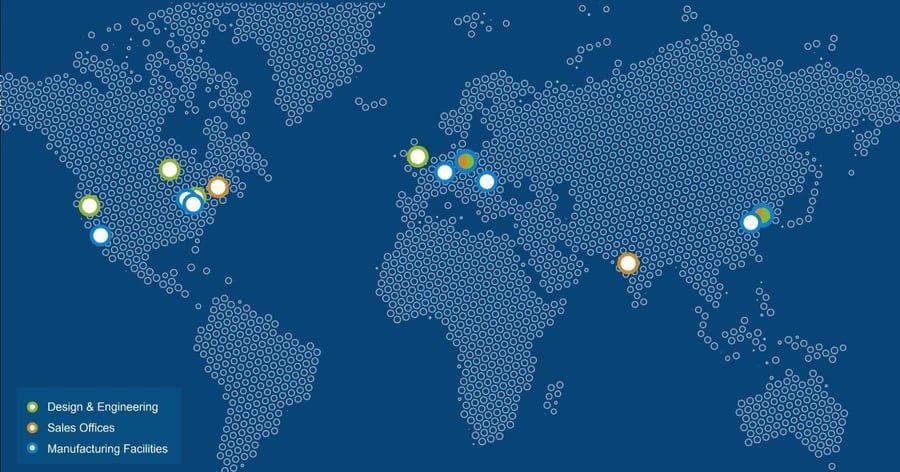
Worldwide production capacity and device development services
Sanner operates a global manufacturing network with a total production space of 28,500 sqm, including 11,000 sqm of cleanroom space (Class 7/8). Our Design Centers of Excellence, Springboard and Gilero, are strategically located in the UK and across the US. Our manufacturing facilities are based in the US, Asia and Europe.
How we can help you
- Capacity for small-batch prototypes and scaling to full production. Our facilities are equipped to handle a wide range of production needs:
- High precision fully electric thermoplastic injection molding capable of handling small shot weights (around 0.1g) up to 64-cavity molds (max. 350T), including 2K molding with thermoplastic elastomers (TPE).
- Manual assembly up to fully automated assembly processes, incorporating mechatronic components for enhanced efficiency and precision.
- End of line packaging for sub-assemblies or finished goods
- Sterilization through trusted external partners, utilizing validated processes to ensure compliance with industry standards.
- Our experience in successfully transferring innovations to production across the EU, US, and China speaks to our ability to bring cutting-edge technologies to global markets. All phases of device development can be supported from our Design and Development Centers of Excellence in both the US and EU.
- Our design transfer processes enable the smoother path to market. We offer end-to-end thinking from development to manufacturing all under one roof to de-risk your project and for a better time to market.
- Our reliability and collaborative working attitude support you in any stage of your project.
Quality control processes and compliance with ISO 13485
Sanner Group facilities are ISO 13485 certified for Design & Development and Manufacturing respectively. Over 250 ISO, AAMI/ANSI standards are used routinely. Our streamlined quality system satisfies the FDA requirements for both the device quality system regulations and the part 210/211 cGMP regulations.